CAS
2023788-19-2
AA-Sequence
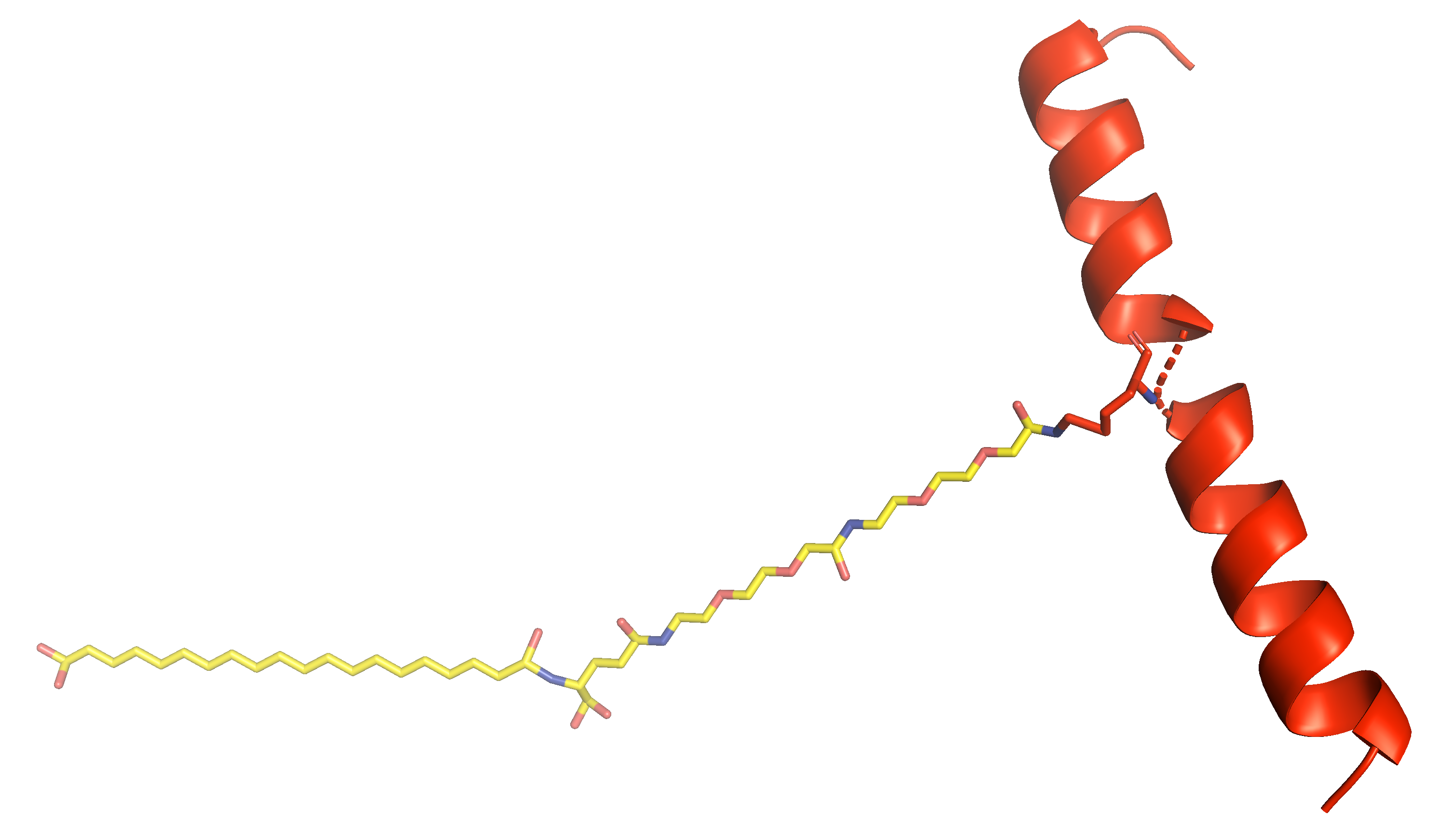
Y-{Aib}-EGTFTSDYSI-{Aib}-LDKIAQ-{diacid-gamma-Glu-(AEEA)2-Lys}-AFVQWLIAGGPSSGAPPPS-NH2
Originator
Eli Lilly and Company
First marketed by
Eli Lilly and Company
Name of FDF
Mounjaro®
Original FDF
RTU solution for subcutaneous injection
Mode of Action
Dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist
Indications
Obesity, type 2 diabetes mellitus
Patent status
Subcutaneous formulation is still patented until 2032-05-13